Hospital Pharmacy Management System Modernization: Why This Is Different From Every Other IT Project
Hospital pharmacy management system modernization carries a risk profile that no other enterprise IT program matches. Medication errors kill approximately 7,000–9,000 US patients annually [1], and a systematic review published in 2025 found that nearly 94% of hospitalized patients experienced at least one prescribing error during admission [2].
The pharmacy system and its drug interaction checks, allergy verification logic, dosing limits, formulary enforcement, and controlled substance tracking is the last automated defense against most of those errors.
A modernization regression in this environment is not a broken feature. It is a potential harm event. That distinction governs every architectural and sequencing decision that follows.
The 24/7 operational constraint makes execution more challenging than in comparable contexts. Hospital pharmacy has no maintenance window. Every modernization approach must account for parallel-run operation, staged cutover by clinical unit, real-time monitoring during transition, and tested rollback protocols as baseline requirements.
Three regulatory mandates are now simultaneously enforced:
- DSCSA: Large dispenser enforcement active as of November 27, 2025; small dispenser enforcement November 27, 2026; no further delays announced
- IRA Maximum Fair Prices: First 10 drugs effective January 1, 2026; 15 more for 2027; 20 more for 2029 onward; MTF data submission requirements active now
- DEA EPCS: Electronic prescriptions for controlled substances expanding across state mandates; legacy systems predating the standard cannot provide auditable digital prescriber verification
The question is no longer whether to modernize. It is how to do it without creating the risk modernization is supposed to eliminate.
DSCSA Is Enforced. IRA Pricing Is Live. What Your Pharmacy System Must Do Now.
DSCSA: An Architecture Problem, Not a Configuration Update
The Drug Supply Chain Security Act requires fully electronic unit-level drug tracing for every covered drug. Not batch-level, lot-level or unit-level, but with GS1-standard serialized product identifiers on every transaction.
Every covered drug transfer must be accompanied by the electronic exchange of Transaction Information, Transaction History, and Transaction Statements. Systems must maintain six years of tracing data and respond to FDA tracing requests within 24 hours. Paper-based systems are explicitly non-compliant under the final rule [3].
Systems built before 2015 were not designed for serialized unit-level tracking. The serialization data model, the TI/TH/TS exchange layer, and the GS1 identifier handling are not features that can be configured into a system built without them. This is an architectural constraint. FDA has already issued warning letters and criminal indictments under active DSCSA enforcement.
Penalty exposure includes civil fines up to $500,000 per violation, criminal charges, including potential imprisonment, license revocation, and FDA authority to confiscate non-traceable products. A shipment that cannot be verified must be quarantined, destroying inventory and generating costly returns. For a high-volume hospital pharmacy, a single enforcement action at that scale is an existential operational event.
IRA/MFP: New Financial Mechanics Your Billing Module Was Not Built For
The Inflation Reduction Act’s Maximum Fair Price mechanism introduces a specific operational requirement: pharmacies dispensing negotiated drugs to Medicare patients must submit claims data to the Medicare Transaction Facilitator to receive rebates. Legacy billing modules were not built for this mechanism [4].
The first 10 drugs carrying Maximum Fair Prices became effective January 1, 2026. The list expands to 15 additional drugs for 2027 and 20 more for 2029 onward, covering high-volume hospital pharmacy categories including diabetes, heart failure, autoimmune, and cancer.
Adam Porath, VP of Pharmacy Services at Renown Health, described the MTF data submission requirement as a significant operational challenge and the financial implications as “concerning,” given uncertainty around how the mechanism will interact with existing contract structures [4].
The 340B interaction compounds the problem. As negotiated Maximum Fair Prices compress list prices, 340B savings margins shrink. The financial modeling that justifies 340B program participation now requires analytics that most legacy pharmacy systems cannot provide. This is addressed in the decision framework below.
DEA EPCS: The Third Forcing Function
Electronic Prescriptions for Controlled Substances are required for Schedule II through V in an expanding set of state mandates. Legacy systems predating the EPCS standard cannot provide the auditable digital prescriber verification that those mandates require.
This is a compliance gap that must be assessed system-by-system; it does not warrant deeper treatment here, but it belongs on the assessment checklist.
Before committing to a path, the right starting point is knowing exactly what your current system can and cannot do. The $0 Modernization Assessment maps your pharmacy system’s compliance capabilities, integration dependencies, and embedded clinical logic and produces a clear recommendation on Path 1, 2, or 3 before you commit. No cost. No obligation.
Path 1, Path 2, or Path 3: How to Make the Right Call for Your Pharmacy System
Vendor sales cycles, regulatory deadlines, and EHR upgrade pressures all create urgency around this decision. None of them creates clarity about what is actually embedded in the current system.
The framework below presents three credible paths with honest tradeoffs, including that Epic Willow is sometimes the right answer. Before any of them, however, is the prerequisite.
The Shared Prerequisite
The decision is only safe to make after a complete inventory of what is embedded in the current system.
Custom integration specifications, drug alert logic tuned to your formulary over years of clinical operation, therapeutic interchange rules, 340B split-billing algorithms, controlled substance thresholds, and the custom workflows that no vendor migration guide accounts for.
Documentation reviews do not produce this inventory. Vendor assessments do not produce this inventory. Codebase archaeology does.
Path 1: Adopt an Integrated EHR Pharmacy Module (Epic Willow / Oracle PharmNet)
Best for: Organizations already on Epic or Oracle Health with minimal custom pharmacy logic, where standard EHR pharmacy workflows are sufficient and native EHR integration is the highest-value outcome.
What you gain:
- Native EHR integration with no custom interface maintenance for order processing, medication reconciliation, or clinical decision support
- DSCSA-capable out of the box, with vendor-managed compliance updates
- Single-platform governance across clinical and pharmacy workflows
- Reduced long-term interface maintenance burden
What you risk losing:
- Years of custom drug alert tuning. Interaction rules and clinical decision thresholds calibrated to your formulary and patient population
- Custom therapeutic interchange rules built to your P&T committee’s specific protocols
- 340B split-billing logic built as a custom workflow on top of the legacy system
- Custom ADC interface specifications for Pyxis or Omnicell configurations unique to your dispensing environment
- Custom clinical reports and workflow automations that clinical staff depend on and that will not migrate automatically
The prerequisite in practice: Before committing to Epic Willow or PharmNet, a complete codebase inventory must exist.
A documented pattern across pharmacy migration programs is that undocumented clinical requirements such as pediatric dosing thresholds, high-alert medication overrides, custom alert suppression logic, surface mid-migration rather than pre-migration.
When that happens, the program either absorbs unplanned scope or goes live with incomplete clinical logic. Neither is acceptable in a dispensing environment.
Vendor context: Oracle Health experienced a significant data breach in February 2025 involving former Cerner servers, with an active FBI investigation. Current Cerner/Oracle clients evaluating Path 1 should assess strategic roadmap continuity and data governance posture before committing.
Path 2: Replace With a Modern Standalone Pharmacy System
Best for: Multi-EHR environments, health systems running both Epic and Oracle Health, or organizations where best-of-breed pharmacy clinical decision support matters more than EHR-native coupling.
What you gain:
- Purpose-built pharmacy clinical decision support, often more sophisticated than EHR-native modules
- Flexibility across EHR environments, with no dependency on a single EHR vendor’s pharmacy roadmap
- Dedicated 340B compliance tooling that integrated EHR modules typically do not match
What you risk:
- Full integration rebuild. Every ADC interface, CPOE connection, BCMA feed, and lab integration must be rebuilt and revalidated from scratch
- Data migration complexity across formulary histories, dispensing records, controlled substance logs, and custom report logic
- Workflow disruption during transition if the cutover is not staged by clinical unit
Vendor context: RedSail Technologies acquired PrimeRx in February 2026, making RedSail the largest US pharmacy software company by count at 16,000 pharmacies [5].
The standalone hospital pharmacy software market, which includes platforms such as Altera Pharmacy (formerly Allscripts), MEDITECH Expanse Pharmacy, and others, is also consolidating. Product roadmap continuity and post-acquisition integration plans must be assessed before committing to any standalone vendor.
A platform decision made today will still be in production in 2030.
Path 3: Modernize the Existing System In Place
Best for: Organizations with heavily customized systems where embedded clinical and compliance logic is extensive. Particularly relevant for safety-net hospitals with complex 340B programs, systems with 20-plus years of custom formulary logic, or organizations where the full interface rebuild cost of Paths 1 and 2 approaches or exceeds the cost of modernizing what exists.
What you gain:
- Embedded clinical logic preserved with full traceability. No reconstruction from documentation, no inferred behavior, no mid-program discovery
- Incremental risk profile: the system stays live throughout, with staged validation by clinical module and rollback capability at every stage
- 340B split-billing algorithms, custom ADC specifications, and therapeutic interchange rules modernized in place, not rebuilt from scratch
- Compliance logic validated against DSCSA and IRA/MFP requirements before go-live, not after
What it requires:
- Full system comprehension before transformation begins; the codebase is the only reliable source of truth for what the system actually does
- An incremental modernization approach that keeps the system live throughout
- Zero-loss handling of formulary histories, dispensing records, and controlled substance logs through any database migration
When it is not the right choice:
- When platform architecture is so degraded that modernization costs approach replacement costs
- When the platform vendor has led to product EOL with no migration path
- When EHR consolidation makes Path 1 structurally inevitable regardless of custom logic volume
What Does Pharmacy System Modernization Cost?
The cost question is one most vendors avoid answering directly. Here is an honest framing by path.
Integrated EHR pharmacy modules like Epic Willow and Oracle PharmNet are not purchased as standalone products. They are components of broader EHR implementations that typically run from $10M to $100M or more, depending on health system size, scope, and existing infrastructure.
The pharmacy module itself is not the primary cost driver; the implementation, training, interface rebuilding, and change management are. Organizations already living on Epic or Oracle Health face a different cost calculus than those considering a full EHR migration to access the pharmacy module.
Standalone pharmacy system replacement runs roughly $100K to $1M-plus for the software and implementation, with variance driven by facility volume, number of dispensing locations, ADC count, and integration complexity. That range does not include the integration rebuild, which is almost always the largest and most consistently underestimated line item, regardless of which path is chosen.
Modernizing an existing system in place is scoped after a dependency assessment, because the cost is driven by codebase complexity, integration volume, and the density of embedded clinical logic, none of which can be estimated accurately from documentation alone. The assessment itself runs in days.
Across all three paths, the integration rebuild cost of rebuilding and revalidating every ADC, CPOE, BCMA, and lab interface is the budget line that most pharmacy modernization programs underestimate at the outset and overrun at the close.
Vendor Comparison: Epic Willow, Oracle PharmNet, and Standalone Platforms
The table below compares the primary decision factors for each path at the platform level. It is a starting framework, not a procurement specification, where each organization’s existing infrastructure, EHR environment, and custom logic volume will shift the calculus.
| Factor | Epic Willow | Oracle Health PharmNet | Standalone Platforms (Altera, MEDITECH, others) |
| Best-fit profile | Epic EHR shops with limited custom pharmacy logic | Oracle/Cerner EHR shops; assess roadmap risk post-breach | Multi-EHR environments; best-of-breed clinical decision support priority |
| DSCSA status | Compliant; vendor-managed updates | Compliant; assess post-acquisition roadmap continuity | Varies by vendor; verify current certification before commitment |
| 340B capability | Basic split-billing support; complex programs typically require a third-party platform | Basic split-billing support; same third-party dependency for complex programs | Dedicated 340B tooling generally stronger than EHR-native modules; verify per vendor |
| ADC integration approach | Native integration with major ADC vendors within Epic ecosystem; custom configurations require rebuild | Native integration within Oracle ecosystem; custom configurations require rebuild | Vendor-specific; full ADC interface rebuild required in all cases |
| Custom logic migration | Requires pre-migration inventory; custom logic does not migrate automatically | Requires pre-migration inventory; same risk as Epic for undocumented logic | Requires full inventory before data migration; no automatic logic transfer |
| Cutover model | Hard cutover; no incremental staging by clinical module | Hard cutover; same risk profile as Epic | Hard cutover; staging possible by location but not by clinical module |
| Vendor stability | High; Epic is privately held with consistent roadmap | Moderate; post-acquisition uncertainty; active breach investigation | Variable; market consolidating rapidly; assess each vendor’s PE backing and acquisition history |
Decision Summary
| Factor | Path 1 (EHR Module) | Path 2 (Standalone Replace) | Path 3 (Modernize In Place) |
| Best fit | Single-EHR shop, minimal customization | Multi-EHR, best-of-breed priority | Heavy customization, complex 340B |
| Integration rebuild | Partial — EHR interfaces native; ADC/BCMA rebuilt | Full rebuild of all interfaces | No rebuild — interfaces preserved and modernized |
| 340B risk | High if custom logic exists in current system | Medium — depends on platform 340B tooling | Low — algorithms preserved with traceability |
| Custom logic preservation | Requires pre-migration inventory to identify what is lost | Requires full inventory before data migration | Inventory-driven; logic preserved by design |
| Cutover risk | High — hard cutover event | High — full replacement | Low — incremental, staged, live throughout |
| Regulatory compliance | Vendor-managed; DSCSA-capable out of box | Depends on vendor; verify before commitment | Validated against DSCSA/IRA requirements during modernization |
| Typical timeline | 12–24 months | 18–30 months | Assessment in days; transformation scope varies |
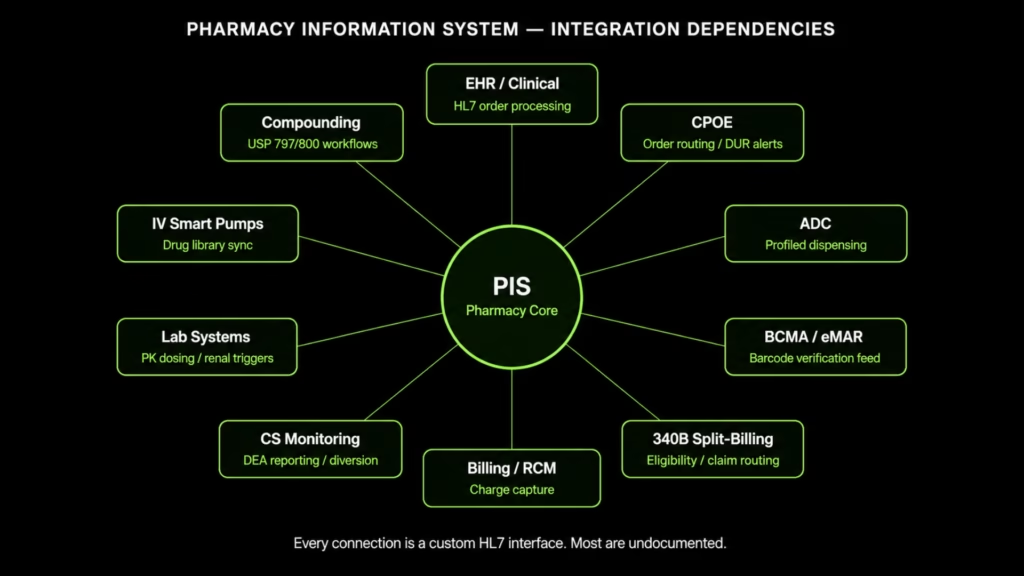
The Integration Layer Problem: 10–15 Connected Systems, Most of Them Undocumented
The decision framework above identifies which path is right in principle. What makes execution of any path harder than it appears is what is described below: the actual scope of what must be rebuilt, revalidated, or preserved.
The Integration Map
Every interface listed below was almost certainly built as a custom HL7 point-to-point connection. It is proprietary, frequently undocumented, and breaks with every system update on either side of the connection.
When the pharmacy system is upgraded or replaced, every interface must be rebuilt and revalidated. That is the most consistently underestimated cost in any pharmacy modernization program.
| Connected System | Integration Type | What Breaks Without It |
| EHR / Clinical System | Bidirectional HL7 order processing, formulary sync, medication reconciliation | Medication orders stop flowing; pharmacist clinical review disconnects from ordering workflow |
| CPOE | Real-time bidirectional order routing and clinical decision support | Prescriber order entry loses formulary enforcement and DUR alerts at point of entry |
| Automated Dispensing Cabinets (ADCs) | Profiled dispensing, controlled substance tracking, override management | Nursing units revert to override mode; pharmacy verification bypassed |
| BCMA / eMAR | Real-time formulary data, NDC cross-reference, barcode verification feed | Bedside scan verification fails or verifies against stale data |
| IV Smart Pumps | Drug library synchronization | Pump drug libraries go stale; dosing limit enforcement degrades |
| 340B Split-Billing Software | Real-time patient eligibility, covered entity status, outpatient claim routing | 340B eligibility determinations made on delayed data; diversion and duplicate discount risk |
| Controlled Substance Monitoring | Dispensing records, DEA reporting, diversion analytics | Regulatory reporting gaps; diversion detection disabled |
| Compounding (USP 797/800) | Admixture workflow, beyond-use dating, lot tracking | Sterile and hazardous compounding workflows lose documentation chain |
| Lab Systems | Pharmacokinetic dosing feeds, renal function adjustment triggers | Clinical pharmacist dosing recommendations lose lab-driven triggers |
| Billing / Revenue Cycle | Charge capture, formulary-to-billing crosswalk, 340B claim routing | Drug charges mis-captured; 340B split-billing logic disconnected from revenue cycle |
ADC Dependency: The Profiled Dispensing Risk
ADCs operate on profiled dispensing: the cabinet releases only what the pharmacy has approved for that specific patient.
When the pharmacy system version mismatches with ADC software, profiled dispensing fails. Nurses move to override mode, which bypasses pharmacy verification entirely. ADC-sourced events account for a significant share of all medication error reports, with a disproportionate fraction involving high-alert medications.
The ADC is only as accurate as the data the pharmacy system feeds it. A version mismatch, a formulary sync delay, or an interface rebuild that goes live before full validation can silently degrade the safety layer that clinical administrators assume is functioning.
BCMA: The Safety Dependency on Current Data
Published evidence establishes that BCMA reduces non-timing medication errors by 41% and potential adverse drug events by 51% when paired with eMAR. That reduction degrades when BCMA is fed outdated or incorrect data from the pharmacy system.
A formulary mismatch, an NDC cross-reference lag, or a post-migration data mapping error can nullify BCMA’s safety benefit for the specific drug records affected, and do so without triggering any visible system alert.
The Drug Database Currency Problem
Specialty drugs now constitute the majority of the drug pipeline, with biosimilar approvals accelerating across oncology, immunology, and rare disease categories. Legacy drug interaction databases are often 12 to 24 months behind current literature.
A pharmacy system missing current specialty drug records from its interaction database is clinically incomplete regardless of its other capabilities, and the gap widens every quarter without active maintenance.
The 340B Integration Layer
For safety-net hospitals, 340B savings are frequently the margin between operational viability and deficit. And the split-billing algorithms, eligibility logic, and audit trail requirements that protect those savings are encoded in pharmacy systems as custom business logic that is almost never documented.
The 340B program permits covered entities such as safety-net hospitals, federally qualified health centers, and qualifying clinics to purchase outpatient drugs at significantly reduced prices. That financial dependency is structural, not secondary.
Compliance requires that the pharmacy system make eligibility determinations (covered entity status, patient eligibility, outpatient vs. inpatient designation) accurately and in real time. Patient status changes affecting 340B eligibility must reach split-billing software on a real-time basis.
Legacy interfaces that batch-update eligibility data are architecturally non-compliant with this requirement. The problem is not a missing feature; it is that the integration model cannot support the timing the program demands.
When eligibility data lags or split-billing logic is misapplied, the result is duplicate discounts or diversion findings at audit. Either outcome triggers repayment obligations, and HRSA audit exposure for covered entities is not theoretical.
Any modernization path that touches the pharmacy system must account for 340B split-billing logic explicitly as a defined pre-migration inventory requirement.
How to Comprehend What Your Pharmacy System Actually Contains Before You Modernize
Every pharmacy migration that surfaces undocumented clinical logic mid-program faces the same two choices: absorb unplanned scope or go live with incomplete logic. Neither is acceptable in a dispensing environment.
The difference between programs that succeed and programs that create new risk while eliminating old risk is whether a full inventory of what the system contains exists before the first transformation decision is made.
A safe pre-migration assessment covers three distinct categories.
Category 1: The Dependency Map
Every EHR interface, ADC connection, BCMA feed, CPOE channel, 340B data flow, controlled substance monitoring module, compounding workflow, lab integration, and billing touchpoint, documented with actual interface specifications, not inferred from vendor documentation that may be years out of date.
This is what makes Path 1, 2, and 3 comparable in actual complexity rather than assumed complexity.
Category 2: The Clinical Logic Catalog
The logic that lives in the codebase and nowhere else:
- Drug alert customizations: interaction rules and thresholds tuned to your formulary and patient population over years of operation
- Therapeutic interchange rules: the specific substitution logic built to your P&T committee’s protocols
- 340B split-billing algorithms: eligibility logic, covered entity routing rules, and outpatient claim handling
- Controlled substance thresholds and DEA reporting workflows
- Custom ADC interface specifications for every cabinet configuration in the facility
In large hospital management system codebases, a significant fraction of business rules, including clinical thresholds hardcoded in modules that have not been opened in years, are undocumented.
Programs that discover these rules mid-migration either absorb unplanned scope or go live with gaps. Programs that inventory them before the program begins do not face that choice.
Category 3: Parity Validation Design
Before transformation begins, the test suite that will validate whether the modernized system produces identical clinical outputs under identical inputs must be defined.
For pharmacy, this means every drug alert rule, every therapeutic interchange decision, every 340B eligibility determination, every controlled substance threshold, every custom report output. Standard QA tests whether a system works.
Clinical parity validation tests whether it works the same way the prior system did, which is the only standard that holds in a dispensing environment.
Legacyleap’s five-agent system executes this methodology across all three categories: dependency mapping, clinical logic extraction, path determination, incremental diff-based transformation with human review at every checkpoint, and parity validation before go-live. The system stays live throughout; rollback capability exists at every stage.
In our engagement with a leading healthtech company (over 300 JSP screens and 5,000-plus Struts forms modernized to Angular in a production 24/7 clinical environment), the operational constraint was identical to hospital pharmacy: the system could not go dark during transformation, and it did not.
Before You Choose Epic Willow, Standalone, or Modernize In Place, Do This First
Every pharmacy migration that surfaces undocumented clinical logic mid-program faces the same two choices: absorb unplanned scope or go live with incomplete logic. Neither is acceptable in a dispensing environment.
The $0 Modernization Assessment maps your system’s dependencies, extracts your 340B and clinical decision support logic, and produces a clear Path 1, 2, or 3 recommendation before you commit. It runs in days. The cost of the assessment is zero. The cost of skipping it is not.
For a broader healthcare application modernization context, including HIPAA compliance framing, see our healthcare application modernization blog.
FAQs
A hospital pharmacy management system (also called a pharmacy information system, or PIS) is the operational core that processes medication orders, enforces formulary rules, manages drug interaction and allergy checking, controls dispensing through ADCs and barcode scanners, tracks controlled substances, and handles 340B split-billing logic. It is not a standalone application. Its data quality directly determines the accuracy of every downstream clinical safety system connected to it.
Large dispenser enforcement became active November 27, 2025. Small dispenser enforcement follows November 27, 2026. No further delays have been announced. The law requires unit-level electronic drug tracing with GS1 serialized identifiers, electronic transaction document exchange, six-year data retention, and a 24-hour FDA tracing response. Systems built before 2015 are unlikely to meet the serialization requirements without significant architectural remediation.
Three categories. Regulatory: pre-2015 systems are architecturally non-compliant with DSCSA and subject to fines up to $500,000 per violation, criminal charges, and license revocation. Clinical: outdated drug interaction databases and ADC interface mismatches degrade the automated safety layers clinical staff rely on. Financial: legacy interfaces that batch-update eligibility data cannot meet 340B real-time compliance requirements, creating direct HRSA audit exposure.
The 340B program allows covered entities to purchase outpatient drugs at significantly reduced prices. For safety-net hospitals, those savings are frequently the margin between operational viability and deficit. Compliance requires real-time eligibility determinations routed to split-billing software without delay. Legacy systems that batch-update eligibility data fail this requirement architecturally. Any modernization program that does not inventory and preserve the 340B logic in the current system before migration creates direct audit exposure.
It depends on what is actually in your current system, and that cannot be known without a codebase-level inventory. Epic Willow or Oracle PharmNet is often right for organizations already on those platforms with limited custom pharmacy logic. Standalone replacement suits multi-EHR environments or best-of-breed priorities. Modernizing in place suits organizations with heavily customized systems or complex 340B programs. The prerequisite for all three paths is the same: a full inventory before any path decision is made.
The test suite must be defined before transformation begins, not assembled after go-live. It needs to cover every drug alert rule, every therapeutic interchange decision, every 340B eligibility determination, every controlled substance threshold, and every custom report output. The standard is not whether the new system works. It is whether it produces identical outputs under identical inputs to the prior system. Discrepancies in drug alert logic or 340B eligibility calculations are clinical and compliance failures, not post-launch QA items.
Profiled dispensing fails when the pharmacy system feed goes out of sync with the ADC software version. When that happens, nurses default to override mode, bypassing pharmacy verification entirely. Prevention requires a complete specification of every custom ADC interface before the upgrade begins, staged validation by the nursing unit before go-live, and a tested rollback protocol. Organizations that treat ADC revalidation as a post-go-live activity encounter the highest-severity incidents consistently.
References
[1] PMC — The role of pharmacists in mitigating medication errors in the perioperative setting: a systematic review. Systematic Reviews, January 14, 2025. https://pmc.ncbi.nlm.nih.gov/articles/PMC11731391/
[2] Frontiers in Digital Health — Using shared clinical decision support to reduce adverse drug events and improve patient safety. November 10, 2025. https://www.frontiersin.org/journals/digital-health/articles/10.3389/fdgth.2025.1703141/full
[3] FDA Official Documentation — Drug Supply Chain Security Act (DSCSA): Final rule, enforcement timeline, and unit-level tracing requirements. https://www.fda.gov/drugs/drug-supply-chain-security-act-dscsa
[4] Becker’s Hospital Review — Adam Porath, VP of Pharmacy Services, Renown Health. “Pharmacy in 2026: How health systems are preparing.” August 25, 2025. https://www.beckershospitalreview.com/pharmacy/pharmacy-in-2026-how-health-systems-are-preparing/
[5] RedSail Technologies — Press release: RedSail Technologies acquires PrimeRx as an affiliate, expanding its network to 16,000 pharmacies nationwide. February 11, 2026. https://www.redsailtechnologies.com/press-releases/redsail-technologies-acquires-primerx-as-an-affiliate